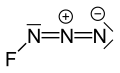Fluorine azide
| |||
| Names | |||
|---|---|---|---|
| Other names triazadienyl fluoride | |||
| Identifiers | |||
CAS Number |
| ||
3D model (JSmol) |
| ||
PubChem CID |
| ||
CompTox Dashboard (EPA) |
| ||
InChI
| |||
| |||
| Properties | |||
Chemical formula | FN3 | ||
| Molar mass | 61.019 g/mol | ||
| Appearance | Yellow-green gas | ||
| Melting point | −139 °C (−218 °F; 134 K) | ||
| Boiling point | −30 °C (−22 °F; 243 K) | ||
| Explosive data | |||
| Shock sensitivity | Extreme | ||
| Friction sensitivity | Extreme | ||
| Hazards | |||
| Occupational safety and health (OHS/OSH): | |||
Main hazards | Extremely sensitive explosive | ||
| NFPA 704 (fire diamond) | [citation needed]  0 4 | ||
| Related compounds | |||
Other cations | Hydrazoic acid Chlorine azide Bromine azide Iodine azide | ||
Except where otherwise noted, data are given for materials in their standard state (at 25 °C [77 °F], 100 kPa). Infobox references | |||
Fluorine azide or triazadienyl fluoride is a yellow green gas composed of nitrogen and fluorine with formula FN3.[1] Its properties resemble those of ClN3, BrN3, and IN3.[2] The bond between the fluorine atom and the nitrogen is very weak, leading to this substance being very unstable and prone to explosion.[3] Calculations show the F–N–N angle to be around 102° with a straight line of 3 nitrogen atoms.[4]
The gas boils at –30° and melts at –139 °C.[5]
It was first made by John F. Haller in 1942.[6]
Reactions
Fluorine azide can be made by reacting hydrazoic acid or sodium azide, with fluorine gas.[5][7]
- HN3 + F2 → N3F + HF
- NaN3 + F2 → N3F + NaF
Fluorine azide decomposes without explosion at normal temperatures to make dinitrogen difluoride:
- 2 FN3 → N2F2 + 2 N2.[1]
At higher temperatures such as 1000 °C fluorine azide breaks up into nitrogen monofluoride radical:[7]
- FN3 → NF + N2
The FN itself dimerizes on cooling.
- 2 NF → N2F2
Solid or liquid FN3 can explode, releasing a large amount of energy. A thin film burns at the rate of 1.6 km/s.[8] Due to the explosion hazard, only very small quantities of this substance should be handled at a time.[9]
FN3 adducts can be formed with the Lewis acids boron trifluoride (BF3) and arsenic pentafluoride (AsF5) at -196 °C. These molecules bond with the first nitrogen atom from the fluorine.[10]
Properties
Spectroscopy
| Parameter | Value[9] | Unit |
| A | 48131.448 | MHz |
| B | 5713.266 | MHz |
| C | 5095.276 | MHz |
| μa | 1.1 | |
| μb | 0.7 |
Shape
Distances between atoms are F–N 0.1444 nm, FN=NN 0.1253 nm and FNN=N 0.1132 nm.[9]
Physical
FN3 has a density of 1.3 g/cm3.[11]
FN3 adsorbs on to solid surfaces of potassium fluoride, but not onto lithium fluoride or sodium fluoride. This property was being investigated so that FN3 could boost the energy of solid propellants.[11]
The ultraviolet photoelectric spectrum shows ionisation peaks at 11.01, 13,72, 15.6, 15.9, 16.67, 18.2, and 19.7 eV. Respectively these are assigned to the orbitals: π, nN or nF, nF, πF, nN or σ, π and σ.[3]
References
- ^ a b Gipstein, Edward; John F. Haller (1966). "Absorption Spectrum of Fluorine Azide". Applied Spectroscopy. 20 (6): 417–418. Bibcode:1966ApSpe..20..417G. doi:10.1366/000370266774386470. ISSN 0003-7028. S2CID 96337253.
- ^ Saxena, P. B. (2007-01-01). Chemistry of Interhalogen Compounds. Discovery Publishing House. p. 96. ISBN 9788183562430. Retrieved 16 June 2014.
- ^ a b Rademacher, Paul; Andreas J. Bittner; Gabriele Schatte; Helge Willner (1988). "Photoelectron Spectrum and Electronic Structure of Triazadienyl Fluoride, N3F". Chemische Berichte. 121 (3): 555–557. doi:10.1002/cber.19881210325. ISSN 0009-2940.
- ^ Peters, Nancy J. S.; Leland C. Allen; Raymond A. Firestone (1988). "Fluorine azide and fluorine nitrate: structure and bonding". Inorganic Chemistry. 27 (4): 755–758. doi:10.1021/ic00277a035. ISSN 0020-1669.
- ^ a b Gholivand, Khodayar; Gabriele Schatte; Helge Willner (1987). "Properties of triazadienyl fluoride, N3F". Inorganic Chemistry. 26 (13): 2137–2140. doi:10.1021/ic00260a025. ISSN 0020-1669.
- ^ Lowe, Derek (21 October 2008). "Things I Won't Work With: Triazadienyl Fluoride". In the Pipeline. Retrieved 15 June 2014.
- ^ a b Benard, D. J.; B. K. Winker; T. A. Seder; R. H. Cohn (1989). "Production of nitrogen monofluoride (a1Δ) by dissociation of fluorine azide". The Journal of Physical Chemistry. 93 (12): 4790–4796. doi:10.1021/j100349a022. ISSN 0022-3654.
- ^ Seder, T.A.; D.J. Benard (1991). "The decomposition of condensed phase fluorine azide". Combustion and Flame. 85 (3–4): 353–362. doi:10.1016/0010-2180(91)90139-3. ISSN 0010-2180.
- ^ a b c Christen, Dines.; H. G. Mack; G. Schatte; H. Willner (1988). "Structure of triazadienyl fluoride, FN3, by microwave, infrared, and ab initio methods". Journal of the American Chemical Society. 110 (3): 707–712. doi:10.1021/ja00211a007. ISSN 0002-7863.
- ^ Schatte, G.; H. Willner (1991). "Die Wechselwirkung von N3F mit Lewis-Säuren und HF. N3F als möglicher Vorläufer für die Synthese von N3+-Salzen = The interaction of N3F with Lewis acids and HF•N3F as possible precursor for the synthesis of N3+ salts". Zeitschrift für Naturforschung B (in German). 46 (4): 483–489. doi:10.1515/znb-1991-0410. ISSN 0932-0776. S2CID 97045269.
- ^ a b Brener, Nathan E.; Kestner, Neil R.; Callaway, Joseph (December 1990). Theoretical Studies of Highly Energetic CBES Materials: Final Report for the Period 2 March 1987 to 31 May 1987 (PDF). Louisiana State University, Department of Physics and Astronomy. pp. 21–27. Archived (PDF) from the original on March 3, 2016. Retrieved 25 June 2014.
External links
 Media related to Fluorine azide at Wikimedia Commons
Media related to Fluorine azide at Wikimedia Commons