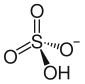
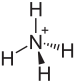Ammonium bisulfate
| |||
 | |||
 | |||
| Names | |||
|---|---|---|---|
| IUPAC name Ammonium hydrogen sulfate | |||
| Identifiers | |||
CAS Number |
| ||
3D model (JSmol) |
| ||
| ChemSpider |
| ||
| ECHA InfoCard | 100.029.332 | ||
PubChem CID |
| ||
| RTECS number |
| ||
| UNII |
| ||
CompTox Dashboard (EPA) |
| ||
InChI
| |||
| |||
| Properties | |||
Chemical formula | (NH4)HSO4 | ||
| Molar mass | 115.11 g/mol | ||
| Appearance | White solid | ||
| Density | 1.78 g/cm3 | ||
| Melting point | 147 °C (297 °F; 420 K) | ||
Solubility in water | Very soluble | ||
| Solubility in other solvents | Soluble in methanol insoluble in acetone | ||
| Hazards | |||
| NFPA 704 (fire diamond) |  3 0 0 | ||
| Safety data sheet (SDS) | External MSDS | ||
| Related compounds | |||
Other anions | Ammonium thiosulfate Ammonium sulfite Ammonium sulfate Ammonium persulfate | ||
Other cations | Sodium bisulfate Potassium bisulfate | ||
Except where otherwise noted, data are given for materials in their standard state (at 25 °C [77 °F], 100 kPa).  N verify (what is N verify (what is  Y Y N ?) N ?) Infobox references | |||
Ammonium bisulfate, also known as ammonium hydrogen sulfate, is a white, crystalline solid with the formula (NH4)HSO4. This salt is the product of the half-neutralization of sulfuric acid by ammonia.
Production
It is commonly collected as a byproduct of the "acetone cyanohydrin route" to the commodity chemical methyl methacrylate.[1]
It can also be obtained by hydrolysis of sulfamic acid in aqueous solution, which produces the salt in high purity:
- H3NSO3 + H2O → (NH4)HSO4
It also arises by the thermal decomposition of ammonium sulfate:
- (NH4)2SO4 → (NH4)HSO4 + NH3
Applications
It can be further neutralized with ammonia to form ammonium sulfate, a valuable fertilizer. It can be used as a weaker alternative to sulfuric acid, although sodium bisulfate is much more common.
Natural occurrence
A related compound of the (NH4)3H(SO4)2 formula, occurs as the rare mineral letovicite, known from coal fire environments.[2][3]
References
- v
- t
- e
| monatomic anions |
|
|---|---|
| oxyanions |
|
| other anions |
|
- Aluminon
- Ammonium acetate
- Ammonium adipate
- Ammonium benzoate
- Ammonium bituminosulfonate
- Ammonium carbamate
- Ammonium citrate
- Ammonium diethyl dithiophosphate
- Ammonium ferric citrate
- Ammonium formate
- Ammonium fumarate
- Ammonium glutamate
- Ammonium lactate
- Ammonium lauryl sulfate
- Ammonium malate
- Ammonium nonanoate
- Ammonium oxalate
- Ammonium picrate
- Ammonium perfluorononanoate
- Ammonium propionate
- Ammonium thioglycolate
- Cupferron
- Ferric ammonium oxalate
- Murexide